Forschungsschwerpunkte
Mammakarzinom:
- Molekulare Marker zur prognostischen und prädiktiven Klassifikation des Mammakarzinoms
- Die Rolle von Adhäsion und Glykosylierung für die Progression und Metastasierung des Mammakarzinoms
- Zerebrale Metastasierung beim Mammakarzinom
- Tumorzelldisseminierung beim Mammakarzinom: Detektion und Charakterisierung von disseminierten Tumorzellen im peripheren Blut und Knochenmark von Patientinnen mit Mammakarzinom
Ovarialkarzinom:
- Molekulare Marker zur prognostischen und prädiktiven Klassifikation des Ovarialkarzinoms
- Untersuchungen zur peritonealen Metastasierung des Ovarialkarzinoms
Zervix- und Vulvakarzinom:
- Klinische Studien zur Prävention, Diagnose und Therapie von HPV-bedingten Krankheiten der Zervix uteri und Vulva
- Klinisch-pathologische Prognosefaktoren und molekulare Marker beim Vulvakarzinom
Projekte im Detail
-
Mammakarzinom
FUNCTIONAL ANALYSIS OF AKT1 FOR THE PROGRESSION AND RADIORESISTANCE IN BRAIN METASTASES OF BREAST CANCER PATIENTS: POTENTIAL ROLE OF AKT INHIBITION AS THERAPEUTIC INTERVENTION
Scientist involved in the project: J. Kempska, K. Legler, A. Grottke, L. Oliveira-Ferrer & V. Müller
Cooperation partner: K. Borgmann, F. Meyer (Laboratory for Radiation Biology & Experimental Radiation Oncology)
Project description: The development of brain metastases is a major limitation of life expectancy in triple negative breast cancer patients. Therapeutic options are limited and systemic treatment options urgently needed.
Our previous findings and recent studies highlight the importance of the AKT/PTEN signaling in the progression and radiosensitization of brain metastases. This suggests that AKT inhibition is a promising therapeutic option and might show synergistic effects in combination with radiation. Using CRISPR/Cas9 technique we have established AKT1 knockout cells, which we used in various experiments (proliferation, migration, invasion, clonogenic assays) to investigate the functional role of AKT1 in the brain seeking breast cancer cell line MDA-MB-231BR. The influence of AKT1 knockout and AKT pharmacological inhibition were also investigated in numerous irradiation experiments. Since new AKT inhibitors (e.g. Ipatasertib) are currently evaluated in the treatment of breast cancer, results from our experiments might be used to guide future clinical trials in the treatment of triple negative breast cancer patients with brain metastases.Funding: Genentech
INFLUENCE OF THE HYALURONIDASE HYAL1 AND THE CD44-HYALURONAN INTERACTION FOR THE DEVELOPMENT OF BRAIN METASTASES IN BREAST CANCER: FINDING NEW POTENTIAL THERAPEUTIC APPROACHES
Involved Scientists from our clinic: F. Hamester, L. Oliveira-Ferrer. I. Witzel, E. Laakmann
Cooperation partner: C. Stürken (Institute of Anatomy und Experimental Morphology)
Project description: Around one-third of all patients with metastatic breast cancer develop brain metastases (BM). Brain metastasis negatively influences the life expectancy and quality of affected patients. Unfortunately, there are still large gaps in our understanding of the mechanisms that lead to BM development and new therapy options are urgently needed.
We have identified hyaluronidase 1 (HAYL1), a catabolic enzyme of the hyaluronan (HA) metabolism, as a marker for BM in breast cancer (Witzel et al. 2017). Based on these data, we currently investigate the molecular mechanisms underlying the development of HYAL1-mediated BM in breast cancer cells. A special focus will be set on the interaction between breast cancer cells with aberrant HYAL1 expression and the blood-brain barrier. To this end, we study the influence of HYAL1 on (A) the adhesion of tumor cells to the brain endothelium, (B) the BBB permeability and (C) the invasive ability of tumor cells through the BBB. Furthermore, the metastasis potential of breast cancer cells with aberrant HYAL1 expression will be investigated in an in vivo mouse model. In addition, the role of CD44, the main HA receptor at the surface of breast cancer cells and essential for HA-dependent signaling, will be investigated during BM development. In this context, blocking CD44 could be an interesting therapeutic option to prevent breast cancer cells from entering the brain.Funding: Werner-Otto-Stiftung & Hamburger Krebsgesellschaft
THE ROLE OF SMAD-SPECIFIC E3 UBIQUITIN PROTEIN LIGASE 1 (SMURF1) IN THE PROGRESSION OF BREAST CANCER: IMPACT ON THE DEVELOPMENT OF BRAIN METASTASIS
Involved Scientists: J. Artzenroth, J. Schwinn, L. Oliveira-Ferrer
Project Description: Brain metastases have a high impact on the life expectancy of patients with breast cancer. Unfortunately, we still don’t have a clear understanding of its underlying mechanisms and we lack effective prognostic markers for its early identification. Our analysis has shown that SMAD Specific E3 Ubiquitin Protein Ligase 1 (Smurf1) expression significantly correlates with diesease progression and the development of brain metastasis in HER2-enriched and triple negative breast cancer patients. Smurf1 itself is an E3-Ubiquin-Ligase with a wide range of molecular interactions and cellular functions. Among others, Smurf1 ubiquitylates RhoA in the cytoplasm, leading to cytoskeleton disruption and an increased rate of breast cancer cell migration and invasion. The aim of this project is to validate Smurf1 as a prognostic marker in breast cancer and to further analyze its specific role on the tumor cell – blood brain barrier interaction and therefore in the development of brain metastasis. Smurf1 knock-down and Smurf1 overexpression in different breast cancer cell lines are planned and the effect on breast cancer cell adhesion to endothelial cells, invasion as well as impact on permeability/integrity of the blood brain barrier will be analyzed.
Funding: Deutsche Krebshilfe
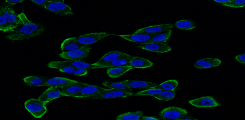
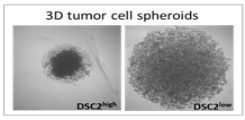
THE ROLE OF THE DESMOSOMAL PROTEIN DESMOCOLLIN-2 IN THE TUMOUR PROGRESSION OF TRIPLE NEGATIVE BREAST CANCERS
Involved scientists from our department: F. Reimer, S. Bryan L. Oliveira-Ferrer
Cooperation partner: C. Stürken (Institute of Anatomy und Experimental Morphology)
Project description: Desmosomes are cell junctions which promote cell-cell contact and stabilise neighbouring epithelial cells. Desmosomal proteins have been associated with tumour cell aggregation, and particularly high plakoglobin expression levels have been found in circulating tumour cell clusters (CTCs) from breast cancer patients. To date, studies focusing on the expression of various desmosomal proteins in a variety of primary cancer types have shown both tumour-suppressive and tumour-promoting effects relating to tumour growth and metastasis. Our research group has recently identified Desmocollin-2 (DSC2) as an unfavourable prognostic marker in triple negative breast cancer (TNBC) patients. Moreover, high DSC2 levels in this patient subgroup is associated with an increased development of lung and brain metastases. In the current project, we aim to elucidate the molecular mechanisms behind these findings. Using genetically modified TNBC cell lines with DSC2high and DSC2low expression levels, along with 3D in vitro models, we are currently analysing the impact of DSC2 on tumour cell proliferation, aggregation and chemosensitivity. Furthermore, we are investigating the associated metastatic potential in a brain metastasis mouse model.
-
Ovarialkarzinom
ANGIOGENESIS PROFILE OF OVARIAN CANCER: IMPORTANCE FOR TREATMENT RESPONSE
Scientist involved in the project: K. Legler, L. Oliveira-Ferrer, C. von Bülow, B. Schmalfeldt
Cooperation partners: M. Alawi (Bioinformatics Core Facility), R. Simon, E. Burandt, G. Sauter (Institute of Pathology)
Project description: Ovarian cancer (OvCa) has the highest mortality among gynecologic tumors, and new therapeutic strategies, especially for platinum-resistant (PR) patients, are urgently needed. Previously, we could identify two angiogenesis-related factors, PDGFRβ and VEGFR2, showing a significantly higher expression in PR-tumors (platinum-resistant tumors) in comparison to therapy responding tumors (platinum-sensitive tumors, Avril et al. Oncotarget 2017), suggesting a notably role of angiogenesis in these group of patients who might therefore benefit from an anti-angiogenic therapy. The recombinant humanized anti-VEGF (vascular endothelial growth factor)-antibody bevacizumab is an angiogenesis inhibitor, approved since 2011 for first-line treatment of advanced OvCa. So far, no predictive marker for response to this VEGF-directed therapy has been described. In the context of the current study we aim to identify a gene signature in OvCa in order to predict platinum-resistance and/or bevacizumab response. In a patient cohort including 244 ovarian cancer tissue samples, gene expression profiles in therapy-resistant vs. therapy-sensitive tumors using transcriptome analyzes (RNA-Seq) were performed and therapy-response signatures were evaluated. In a next step, potential candidate genes/signatures will be validated immunohistochemically.
Funding: Wilhelm-Sander Stiftung
MOLECULAR CHARACTERISTICS AND TUMORIGENICITY OF ASCITES-DERIVED TUMOR CELLS IN OVARIAN CANCER: MITOCHONDRIA METABOLISM AS A NOVEL THERAPY TARGET IN OVARIAN CANCER
Scientist involved in the project: Y. Ding, L. Oliveira-Ferrer, B. Schmalfeldt
Cooperation partners: V. Labitzky, C. Stürken (Institute of Anatomy und Experimental Morphology)
Project description: Ovarian cancer (OvCa) disseminates principally in the peritoneal cavity. Here, detached tumor cell aggregates (spheroids) from the primary tumor are regarded as “metastatic units”, which exhibit a low sensitivity to classical chemotherapy, probably due to their unique molecular characteristics and the influence of the ascites microenvironment. The high metastatic potential of these cell-aggregate structures could be confirmed by our group, where ascites-derived spheroids (>30µm) from high-grade serous OvCA (HGSOC) patients -which were confirmed tumor cells-enriched by flow cytometry (EpCAM+/CD24+)- successfully develop peritoneal carcinosis in an intraperitoneal mouse model. Using RNAseq analysis of ascites-derived tumor cells versus tumor tissue-derived cells, we identified several deregulated factors and cellular pathways that might be responsible for the high metastatic potential and the low chemotherapy sensitivity of ascites-spheroids. Currently, our studies aim to validate the potential role of these factors as new potential treatment options in advanced ovarian cancer.
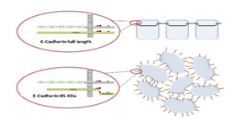
THE FUNCTIONAL RELEVANCE OF E-CADHERIN FRAGMENTS IN OVARIAN CANCER PROGRESSION
Scientist involved in the project: K. Legler, S. Kürti, L. Oliveira-Ferrer, B. Schmalfeldt
Project description: In Epithelial Ovarian Cancer (EOC), metastasis occurs mainly intraperitoneal and retroperitoneal, while extra-abdominal metastases are a rarity. So far, the molecular mechanisms of the two main metastatic pathways are poorly understood. The cell adhesion molecule E-cadherin and precisely its fragment E-cadherin-85kDa could be identified as a molecular marker for intraperitoneal metastasis in EOC (Trillsch et al. Br J Cancer 2016). In the context of the actual project we aim to gain further insight into the Calpain-mediated E-cadherin fragmentation, and its modulation as a possible therapeutic target for the treatment of metastatic EOC. Using two calpain inhibitors, functional assays such as tumor cell proliferation, migration and chemosensitivity are performed in vitro. Additionally, we examine the impact of Calpain inhibition on tumor progression and metastasis in an intraperitoneal ovarian cancer mouse model.
Funding: Erich und Gertrud Roggenbuck-Stiftung
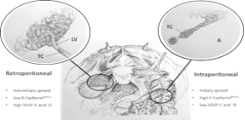
INTRA- UND RETROPERITONEAL METASTASIS IN OVARIAN CANCER: TWO BIOLOGICALY DIFFERENT TUMOR TYPES?
Scientist involved in the project: K. Legler, S. Kürti, A. Volk, L. Oliveira-Ferrer, B. Schmalfeldt
Project description: Epithelial Ovarian Cancer (EOC) disseminates mainly intraperitoneal and/or retroperitoneal (lymphatic), while distant metastases are rarely seen. So far, the biologic background of these two different modes of progression is poorly understood. Recent studies have shown that the EMT (epithelial-mesenchymal-transition) stage may influence the route of metastases in ovarian cancer. Our group could show that strong tumor expression of the 85 kDa E-Cadherin fragment is associated with intraperitoneal metastasis in EOC, whereas high levels of VEGF-C and VEGF-D correlate with retroperitoneal tumor dissemination. VEGF-C and –D are members of the vascular endothelial growth factor (VEGF) family and promote lymphangiogenesis by binding their cognate receptor VEGFR3 on lymphatic endothelial cells. We could also show that high levels of angiopoietin-2 (Ang2) are associated with retroperitoneal metastasis in EOC. Angiopoietin-2 is a vascular growth factor that play a role in angiogenesis. Targeting Ang2 via specific blocking antibodies has been shown to decrease metastasis to lymph nodes. In the context of this project we aim to establish molecular markers that help us identify patients with high risk of lymphatic metastases, who might benefit from systemic lymphadenectomy and specific antiangiogenic therapy.
COMPARATIVE ANALYSIS OF 3D-MICROTUMORS OF OVARIAN CANCER PATIENTS AND FUNCTIONAL IN VITRO TESTING OF IMMUNOTHERAPEUTIC APPROACHES
Involved scientists of the clinic: Y. Ding, L. Hell, L. Oliveira-Ferrer
Cooperation partners: T. Sturmheit (2cureX GmbH), J. Wellbrock, W. Fiedler (II. Medical Clinic)
Project description: Every year almost 10.000 women are diagnosed with ovarian cancer in Germany. Since most patients are diagnosed at advances stages and recurrence of disease is frequent, the 5-year-survival rate is currently below forty percent. Debulking surgery followed by adjuvant chemotherapy constitutes the standard of care but alternative treatment options, such as immune checkpoint therapy, are already being evaluated in (pre-)clinical trials. Within the realm of this project, 3D-microtumors derived from solid tumor tissue and ascites of ovarian cancer patients are compared regarding their cellular composition, plasticity and protein expression of checkpoint molecules with potential immunotherapeutic relevance, e.g. PD-L1, TIGIT ligands. By doing so, we will gain a deeper understanding of relevant target structures for immunotherapeutic interventions. Additionally, we are planning to measure, quantify and compare the individual responses of patient-derived microtumors to different checkpoint inhibitors upon co-culture with autologous immune cells from solid tumor tissue, ascites or peripheral blood. Ultimately, we aim to develop a functional assay system for pre-therapeutic testing of checkpoint inhibitors and combinatorial approaches for patients suffering from ovarian cancer.
Funding: BMBF KMU-innovativ: Medizintechnik
-
Vulvakarzinom und Zervixkarzinom
11. Klinische Studien zur Prävention, Diagnose und Therapie von HPV bedingten Krankheiten der Zervix uteri und Vulva
Beteiligte Wissenschaftler der Klinik: L. Wölber, L. Kock, A. Runge
Kooperationspartner: Industriepartner, Institut für Pathologie
Projektbeschreibung: Im Rahmen der "Dysplasiesprechstunde" der Klinik für Gynäkologie beschäftigen wir uns mit Diagnostik, Therapie und Nachsorge von Erkrankungen des Muttermunds, der Scheide sowie des äußeren Genitaltrakts. Diese Erkrankungen werden häufig durch humane Papillomaviren (HPV) verursacht, wobei sogenannte "low-risk"-HPV-Typen vor allem genitale Kondylome (Feigwarzen) verursachen, während die "high-risk"-HPV-Typen sog. Dysplasien (präkanzeröse Läsionen), hervorrufen können, welche zu einem geringen Prozentsatz zu einem Krebs entarten können. In den letzten Jahren wurden gegen die häufigsten dieser HPV-Typen Impfstoffe entwickelt, welche im Rahmen multizentrischer Studien in unserer Klinik erprobt werden . Hierzu gehört neben der Impfung auch die regelmäßige Nachuntersuchung geimpfter Patientinnen, um das Auftreten möglicher HPV-induzierter Läsionen zu überprüfen. Daneben werden im Rahmen weiterer Studien neue Methoden zur Optimierung der Behandlung bereits vorhandenen Dysplasien und genitaler Kondylome erprobt.
Drittmittel: Industriepartner
12. Klinisch-pathologische Prognosefaktoren und molekulare Marker beim Vulvakarzinom
Beteiligte Wissenschaftler der Klinik: L.Wölber, K. Prieske, L. Oliveira-Ferrer
Kooperationspartner: Institut für Pathologie, Institut für Tumorbiologie
Projektbeschreibung: Unsere Klinik besitzt langjährige Erfahrung auf dem Gebiet der operativen und konservativen Therapie des Vulvakarzinoms. Im Vergleich zu anderen Klinken in Deutschland wird bei uns jedes Jahr eine hohe Zahl von Patientinnen mit dieser seltenen Krebserkrankung behandelt. So stehen uns sowohl eine große Anzahl von Gewebeproben als auch operative Daten und klinische Verläufe zur Verfügung. Wir beschäftigen uns zum einen mit rein klinischen Fragestellungen, wie der Wertigkeit verschiedener operativer Verfahren (z.B. Sentinelmethode) zum anderen auch mit molekularen Markern des Vulvakarzinoms. Ausgehend von der Tatsache, dass normale Regulationsmechanismen, die Zellwachstum und die Zelldifferenzierung steuern, in bösartigen Tumorzellen ausgeschaltet sind oder umgangen werden, untersuchen wir mit Hilfe der vorhandenen Ressourcen mögliche prognostische und prädiktive Faktoren. Hierzu gehören z.B. Zellzyklus-regulierende Faktoren und Proteine der intrazellulären Signaltransduktion. Die Expression dieser Faktoren wird auf Protein-Ebene analysiert und die Ergebnisse mit den vorhandenen klinisch-pathologischen Parametern korreliert. Um profundere Ergebnisse zu Therapieeffektivität und Prognosefaktoren des Vulvakarzinoms anhand großer Kollektive erreichen zu können, haben wir uns außerdem mit mehreren deutschen Zentren zusammengeschlossen, um gemeinsam eine große Anzahl von Patientinnen anonymisiert zu untersuchen (CaRE Studie).